Taking an environmentally sensitive approach to pest management
Cedar-Quince Rust
Co-authored by
Oscar Pérez-Hernández, Assistant Professor–Crop Protection, Northwest Missouri State University
Francisco Sautúa, Visiting Ph.D. student, University of Buenos Aires, Argentina
Kaitlyn Bissonnette, Assistant Extension Professor-Plant Pathologist, University of Missouri Columbia
Published: September 5, 2018
What is cedar-quince rust?
Cedar-quince rust is a disease caused by a fungus called Gymnosporangium clavipes, which can infect about 480 species in the rose family, including apples, mountain ash, flowering quince, and hawthorn, and members of the Cupressaceae family such as the eastern red cedar (also known as red juniper). Cedar-quince rust causes flagging of leaves (Figure 1), decay and eventually death of the trees (Figure 2), particularly after repeated, untreated infections.

Figure 1 Eastern red cedar (Juniperus virginiana) trees severely affected by the cedar-quince rust. Circle shows a patch of dead leaves (flagging) on the tree. [Photo taken in Maryville, MO. May 2018]

Figure 2 Eastern red cedar trees killed by the cedar-quince rust. [Photo taken in Maryville, MO. May 2018]
Life cycle
The cedar-quince rust fungus requires two distinct plant hosts to complete its life cycle. One host is called the primary host and the other the alternate host. Primary hosts are often different species of evergreens, such as junipers (Juniperus spp.) and red cedars, whereas alternate hosts include the flowering quince, apple, hawthorn and crabapple, among others. On both types of hosts, the fungus produces at least four different types of microscopic spores in specialized structures or fruiting bodies. One type are the teliospores, which are formed in structures called telia and are responsible for the infections in the spring and summer on evergreen hosts (Figure 3).
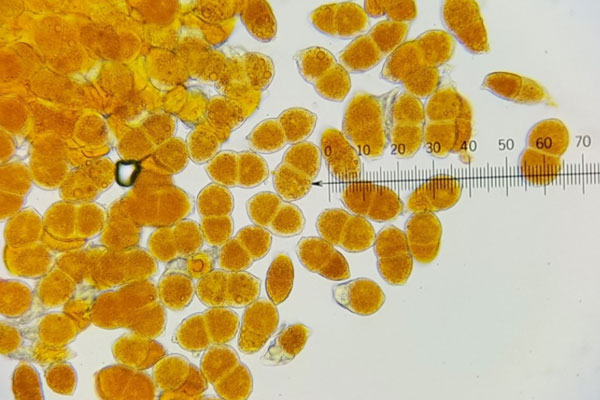
Figure 3 Teliospores of the cedar-quince rust pathogen (G. claviceps) observed under a compound microscope at 400x magnification. [Photo: Sautua and Perez-Hernandez]

Figure 4 Cedar-quince rust aecia on hawthorn (Crataegus spp.) fruit. [Photo by Dr. Alan Windham]
Infection on evergreen hosts occurs on needles, twigs and branches causing swellings that are the developing telia. Development of teliospores within the telia (Figure 3) is favored by moist and cool temperatures (around 76°F). Upon maturity, teliospores germinate in situ (where they are formed) and produce another type of spore called a basidiospore that can only infect plants in the Rosaceae family. Basidiospores are released and carried by wind and rain splash to newly emerging leaves of deciduous hosts. Infection occurs when these spores land on a susceptible plant host and penetrate host cells. Seven to ten days after infection, spots or swellings develop on the infected plant tissue followed by the formation of the next type of spores called spermatia (pycniospores) in structures called pycnia (spermogonia). Spermatia are visible as tiny black dots within large spots. Four to seven weeks later, aeciospores are formed inside tubular protective sheaths called aecia (Figure 4). Aeciospores are released during rain, taken by wind currents over long distances and infect susceptible evergreen hosts during fall and late summer. After germinating on a Juniperus host, an overwintering latent mycelium is produced. The telial state appears on Juniperus in the spring to begin the life cycle again.

Figure 5 Telia erupting from infected cedar tissue immediately following a warm rain. [Photograph credit: Mike Boehm, Dept. Plant Pathology, The Ohio State University, published in APS Image Resources [https://www.apsnet.org/publications/imageresources/Pages/IW000013.aspx]
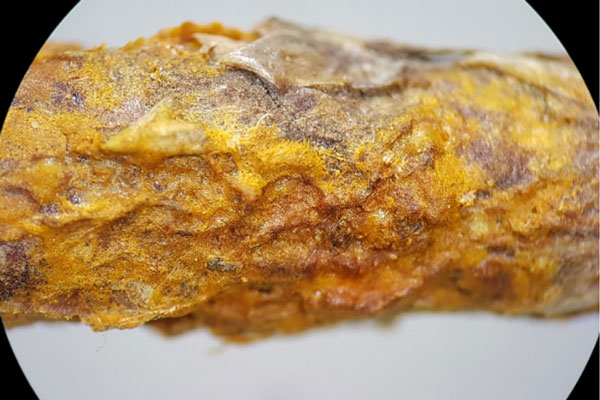
Figure 6 Closed up of a twig with orange dry blisters already passed the gelatinous stage of the fungus and covering the entire surface of the twig. [Photo: Sautua and Perez-Hernandez]

Figure 7 Close up of orange telial blisters of cedar-quince rust on an eastern red cedar branch. The orange areas are the dried out pustules of the fungus that had already broken through the surface. [Photo: Sautua and Perez-Hernandez]
Symptoms and Recognition
Cedar-quince rust symptoms vary according to the infected host plant. On primary hosts (evergreens such as junipers and red cedars), symptoms typically appear as elongated swellings on branches and twigs. Those swellings contain the teliospores that protrude from the plant surface as a bright orange gelatinized matrix (Figure 5). Then the swellings dry out after a few days (Figure 6), break through the surface and cause dieback (Figure 7). While some affected branches and twigs are killed, others can remain infectious for many years (perennial swellings).
On alternate hosts (deciduous hosts such as apples and hawthorn), the fungus infects leaves, petioles, young stems, branches and fruit. The main aecial host is quince (Cydonia oblonga), but apples (Malus pumila) and hawthorn (Crataegus spp.) also are infected. Young branches and fruit are infected more frequently than leaves. Pycnia and aecia may form in fruits in certain hosts. On hawthorn, the aecia look like pinkish tubes that occur mainly on branches, thorns, and fruit (Figure 8).

Figure 8 Cedar-quince rust aecia on hawthorn (Crataegus spp.) fruit. [Photo by Damon Brunette (https://mushroomobserver.org/image/show_image/153998?obs=70212)].
How can cedar-quince rust be recognized? The disease is generally easy to identify at the telial and aecial stages given the symptoms and signs that are produced. Follow the symptom description and photos provided in this publication to help you identify cedar-quince rust. To confirm the diagnosis, consult your regional agronomist at your local extension office, the Plant Pathology Extension Specialist at the University of Missouri or the MU Plant Diagnostic Clinic.
Management
Control of this disease is difficult and must be addressed with an integrated management plan that includes:
Cultural control: keep the trees healthy through proper nutrition and water supply, especially during months of dry weather. Avoid overhead irrigation in the early spring in the afternoons, as high moisture favors rust spore germination.
Resistant varieties: There is limited information regarding resistant cultivars of eastern red cedar to cedar-quince rust, yet some apple cultivars are reported to have some level of resistance to this disease. Consider using resistant cultivars of the alternate hosts if they are planted near the primary host.
Sanitation: Eliminate infected plant material of alternative hosts in the fall. Pruning out branches and twigs with cedar-quince swellings and cankers in early spring helps reduce the amount of inoculum. It is recommended to remove as much of the infected twigs, branches, leaves and fruit as possible and destroy the residue. This practice is helpful, yet does not entirely solve the problem. If trees are severely infected or killed, then remove them and replace them with other ornamental trees that are adaptable in the region.
Fungicides: Fungicides can be applied as a preventive measure to protect developing twigs and branches from infection. On primary hosts, the best time to apply a fungicide is at the beginning of the spring, when plants start to grow actively. Treatments could still provide good results when applied before the swellings become gelatinous. Otherwise, applications when the swellings become dry and break through the bark are less effective. Fungicides registered for use include Captan, Chlorathalonil (Daconil), Mancozeb, Sulfur, Thiram, and Ziram. Refer to local university extension office, plant pathologist, or arborist for fungicide recommendations. Always read and follow the label directions before using a fungicide.
Additional information
A few rust diseases related to the cedar-quince rust occur in the Midwest: the cedar-apple rust and cedar-hawthorn rust. For information on these rust diseases visit the following sites:
- Cedar-apple rust:
http://www.missouribotanicalgarden.org/gardens-gardening/your-garden/help-for-the-home-gardener/advice-tips-resources/pests-and-problems/diseases/rusts/cedar-apple-rust.aspx
http://msue.anr.msu.edu/news/cedar_apple_rust_a_tale_of_a_fungal_disease_with_two_hosts - Cedar hawthorn rust:
http://www.missouribotanicalgarden.org/gardens-gardening/your-garden/help-for-the-home-gardener/advice-tips-resources/pests-and-problems/diseases/rusts/cedar-hawthorn-rust.aspx
http://extension.illinois.edu/focus/index.cfm?problem=cedar-hawthorn-rust
Sources:
Douglas S. M. 2010. Gymnosporangium rusts: common cedar rust diseases in Connecticut. The Connecticut Agricultural Experiment Station.
Himelick, E.B.; Neely, D. 1960. Juniper hosts of cedar-apple and cedar-hawthorn rust. Plant Disease Reporter 44:109-112.
http://www.missouribotanicalgarden.org/gardens-gardening/your-garden/help-for-the-home-gardener/advice-tips-resources/pests-and-problems/diseases/rusts/cedar-quince-rust.aspx. Retrieved on July 7, 2018.
Brand B, Brand G, Shattock R (2006) Sorting out gymnosporangium species = The aecial stage. Field Mycology 74: 123-127.
Kern F.D. 1973. A revised taxonomic account of Gymnosporangium. University Park, PA: Pennsylvania State University Press. 134 p.
Neely, D. 1983. Chemical control of cedar-apple and cedar hawthorn rusts. Journal of Arboriculture 9:85-87.
Subscribe to receive similar articles sent directly to your inbox!
- Cedar Apple Rust (05/02/19)
- Cedar-Quince Rust (09/05/18)
- Elderberry Rust Defoliating Plants (04/27/17)
REVISED: February 21, 2017